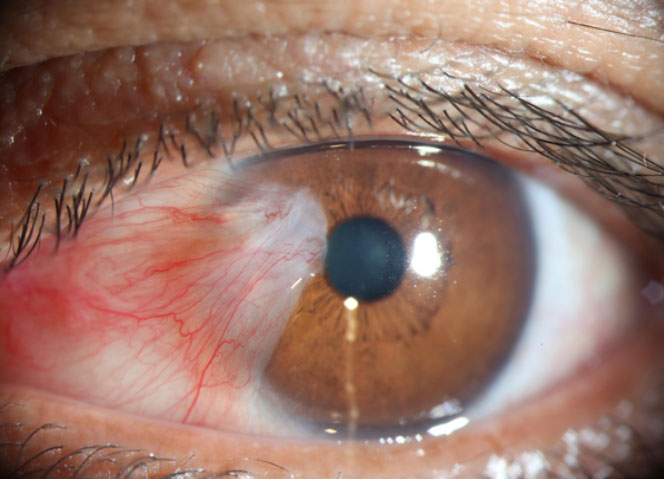
Pterygium
Pterygium is a wing-shaped, fibrovascular lesion on the ocular surface that usually originates from the bulbar conjunctiva and extends onto the corneal limbus and beyond. As the disease progresses, the lesion distorts the corneal topography, encroaches on the visual axis, and obscures the optical center of the cornea, causing visual impairment. The pathogenesis of pterygium is mostly unknown; however, multiple mechanisms have been proposed. These include UVB exposure and release of platelet-derived growth factor (PDGF), overexpression of defensins and phospholipase D, mutations in Kras, p53/p63, and increased levels of VEGF, bFGF, cytokines, and MMPs
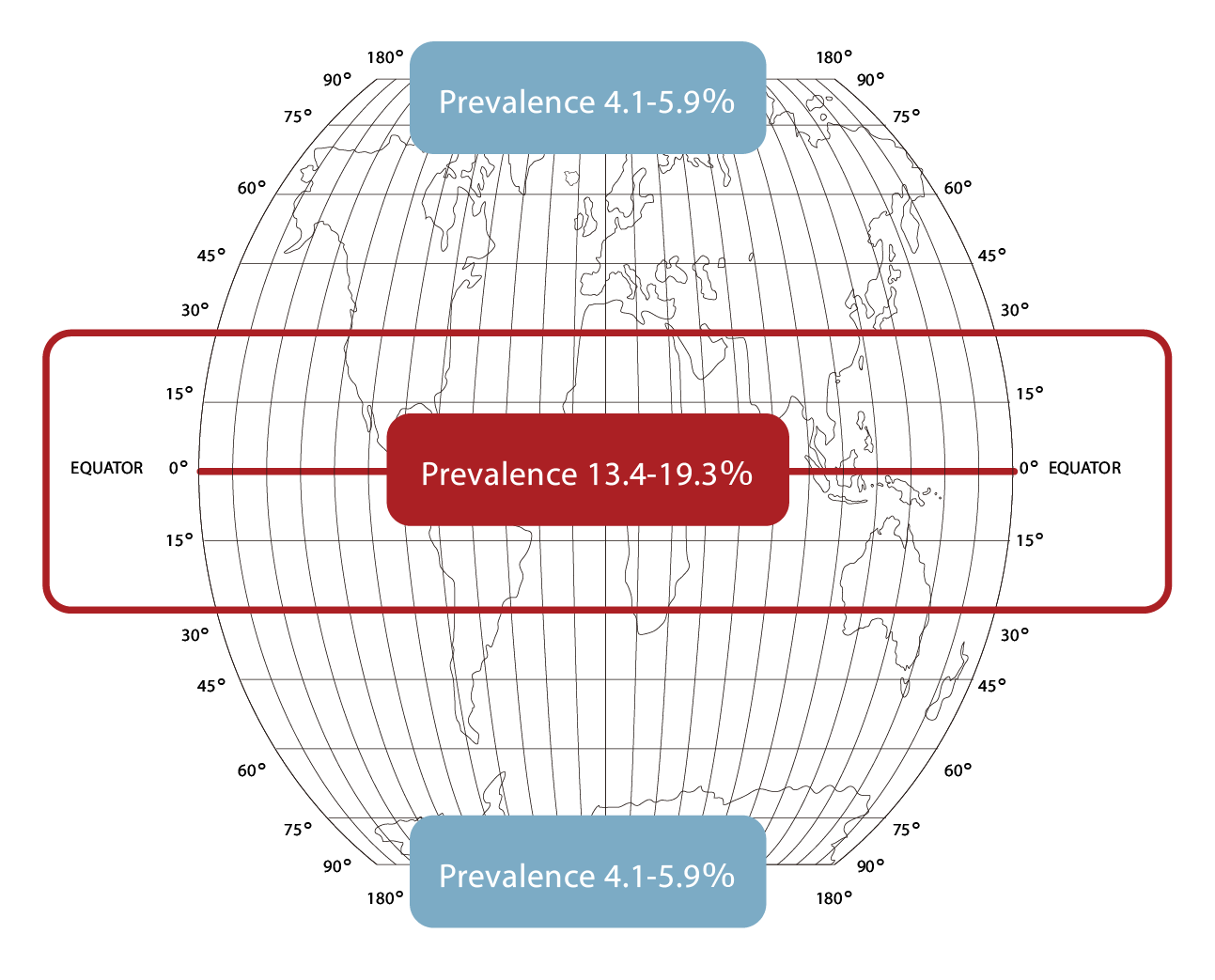
- The total number of patients with hyperemia associated with primary and recurrent pterygium, who are eligible to receive topical treatment is estimated at 3.3 mil in US, EU-5, JP, AU, TW and SG.
- The addressable market of AG-86893 is about 15% (50% of compliance and 30% of penetration).
Treatment Options for Pterygium are Limited
Currently, there is no approved drug for pterygium on the market. Doctors may provide products for symptom relief, such as tear substitutes, topical steroids to reduce inflammation, and recommendations to wear sunglasses to reduce UV exposure and decrease growth stimulus. When vision is impaired by the pterygium, patients may undergo surgery to remove the lesion.